
What is in your water?

CALCIUM
is a naturally occurring mineral necessary for the human body. It is considered one of the six essential minerals. It must be incorporated into the diet through outside sources, such as dairy products, different fish species, spinach and broccoli among other sources.

MAGNESIUM
is a mineral belonging within the alkaline earth metals. Magnesium plays a central role in metabolic, muscular, and cardiovascular performance.

SODIUM
is one of the chemical elements found in table salt. Salt, also known by its chemical name Sodium Chloride, is a crystal-like compound that is abundant in nature and is used to flavor and preserve food. The US government recommends limiting daily sodium intake to 2,300 milligrams (one teaspoon) because too much sodium increases the risk for developing high blood pressure, heart disease and stroke.
Some bottled waters can be high in sodium. The Food and Drug Administration allows waters with less than 5mg of sodium per 360mL of water (Reference Amount Customarily Consumed) to claim they are “Sodium Free”. This means 13.89mg of sodium per litre of water.
SPARKLING WATER |
SODIUM
|
MAGNESIUM
|
CALCIUM
|
ALKALINE
|
|---|---|---|---|---|
AQUA Carpatica Sparkling |
4.4 |
77.0 |
260.0 |
6.6 |
Poland Spring Sparkling |
9.8 |
1.4 |
8.3 |
4.7 |
Perrier |
10.0 |
4.5 |
160.0 |
6.0 |
San Pellegrino |
34.0 |
53.0 |
160.0 |
5.6 |
Gerolsteiner |
130.0 |
108.0 |
348.0 |
6.12 |
Apollinaris |
480.0 |
120.0 |
90.0 |
5.8 |
STILL WATER |
SODIUM
|
MAGNESIUM
|
CALCIUM
|
ALKALINE
|
|---|---|---|---|---|
AQUA Carpatica Still |
ND |
17.0 |
52.0 |
8.2 |
Acqua Panna |
7.0 |
6.7 |
32.0 |
8.2 |
Evian |
6.9 |
27.0 |
81.0 |
7.2 |
Poland Spring Still |
7.3 |
0.69-1.6 |
4.0-11.0 |
6.5-7.9 |
Volvic |
12.0 |
8.3 |
12.0 |
6.92 |
Fiji |
18.0 |
15.0 |
19.0 |
7.9 |
Source: NSF International Lab Test Report 2015, and brand websites
What is natural sparkling mineral water?
Natural sparkling mineral water occurs in select mineral water springs with natural emissions of Carbon Dioxide (CO2) and a significant content of mineral salts, such as Calcium Carbonate (CaCO3) and Magnesium Carbonate (MgCO3), from the geological structures surrounding the source. In volcanic aquifers where mofetic CO2 is present, the CO2 combines with the water, creating a natural sparkling mineral water.

Natural sparkling mineral water and your health
Consuming a naturally sparkling mineral water with natural bicarbonates
neutralizes excessive gastric acidity, stimulates digestion and metabolism, and delivers natural electrolytes to the human body.
Other benefits include
preventing kidney stones and neutralizing lactic acid
generated during physical activity.
Naturally Sparkling
– NATURAL MINERALS
– SLOW PROCESS
– IDEAL ACIDITY IN THE STOMACH
– NATURAL BICARBONATES
VS
Soda Water
– NO MINERALS
– AGRESSIVE PROCESS
– HIGH LEVEL OF ACIDITY IN THE STOMACH
Natural sparkling water is not the same as soda water.
When it comes to health, they act differently on the body.
Conversely, artificially carbonated soda waters contain no natural electrolytes (HCO3).
The artificial addition of CO2 can create a harsh environment throughout the digestive system
by generating Carbonic Acid in the stomach.
This increases gastric acidity, which may ultimately lead to ulcers, acid reflux, hypertension, and others.
Soda water may also have a bitter, metallic, or sour aftertaste.
Naturally Sparkling Mineral Water
Natural Impregnation
(Underground Hydrogeological Aquifer)
Calcium Bicarbonates
Magnesium Bicarbonates
Potassium Bicarbonates
Calcium, Magnesium and Potassium
Bicarbonates present in natural sparkling mineral water increase its
alkalinity
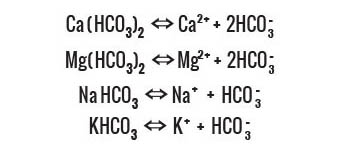
Digestion Process
The increased
alkalinity
buffers excess acidity in the stomach
Soda Water
Artificial Impregnation Process
(Bottling Line)
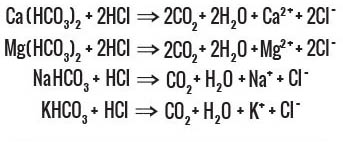
Carbonic acid H2CO3 resulting from artificial impregnation of water with CO2 is unstable and highly acidic
Consumption Process
There are no cations (minerals) to form bicarbonates. The carbon dioxide (CO2) is released aggressively, increasing gastric acidity. This may cause stomach distention, acid reflux, ulcers, and hypertension.
AQUA Carpatica: Naturally Alkaline Spring Water (Still)
The high pH value (8.2) of AQUA Carpatica still water is naturally alkaline
due to the presence of bicarbonate ions in the aquifer of the spring.

Nature’s Process
The contact of water with subterranean carbonate rocks of the Carpathian Mountains causes the following reactions to take place:
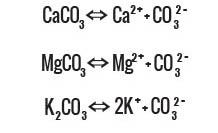
Carbonate ions CO2-3 hydrolyse as follows:
Digestion Process
This enriches the water with a significant quantity of bicarbonates (HCO2-3)
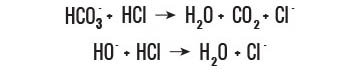
Bicarbonates are beneficial for a healthy digestion. A high bicarbonate concentration increases the buffer capacity of AQUA Carpatica still water. This means that during digestion, the presence of the bicarbonate anions and hydroxyl decreases the excess of acidity in the stomach.
SPARKLING WATER |
ALKALINITY
|
|---|---|
AQUA Carpatica Sparkling |
6.6 |
Poland Spring Sparkling |
4.7 |
Perrier |
6.0 |
San Pellegrino |
5.6 |
Gerolsteiner |
6.12 |
Apollinaris |
5.8 |
STILL WATER |
ALKALINITY
|
|---|---|
AQUA Carpatica Still |
8.2 |
Acqua Panna |
8.2 |
Evian |
7.2 |
Poland Spring Still |
6.5-7.9 |
Volvic |
6.92 |
Fiji |
7.9 |

